AN INTRODUCTION TO THE (MACRO) ALGAE OF
BRITISH COLUMBIA

Cymathaere triplicata, photo by Michael Hawkes.
by Colin Bates
Department of Botany
University of British Columbia
To read the entire PDF of this article, click here.
To visit the E-Flora BC algae atlas pages, click here.
Many people are unaware that British Columbia is a veritable hot spot of seaweed biodiversity. At last count, there were approximately 650 macroalgal species found in waters of the Pacific Northwest (Scagel et al. 1989, Gabrielson et al. 2000). Of these, 530 are known to occur in B.C.!
Why are so many people unaware of the diversity of seaweeds that inhabit British Columbia's waters and shores? Many initial encounters with seaweeds can be unpleasant, especially if you are wading your way into the ocean for a swim, or if you are trying to maintain your footing while scrambling around on the rocks. As well, detached seaweeds end up washing ashore and amassing into smelly decomposing piles. Seaweeds generally get a bad reputation in the press (if any at all). Books like "Killer Algae" (Meinesz, 1999), which describes the invasive nature of a green seaweed species called Caulerpa taxifolia, and talk of 'Harmful Algal Blooms" (a microalgal phenomenon) do not help! All of these factors contribute to the idea that seaweeds are a nuisance. Yet, it is my experience that seaweeds are most certainly not a nuisance, and instead are beautiful organisms with fascinating natural history, diverse economic uses, and essential ecological roles. I hope by the end of this article that you will agree!
To begin our discussion, it is important to
know that seaweeds are a subset of a larger group known as the
algae. It is therefore useful to start off with a short discussion
about the nature of this larger group.

Sea Staghorn (Codium fragile), photo by Colin Bates
What are algae?
When most people think of algae, they conjure a green scum on the side of a pond or fish tank, but in reality algae can be large or small, helpful or harmful, and believe it or not, stunningly beautiful and head-scratchingly interesting.
Unlike plants and animals, the algae do not arise from a single common ancestor (a condition referred to as monophyly. Instead, they are a group composed of many lineages (they are polyphyletic; Graham & Wilcox 2000). Throughout the history of algal taxonomy, various characters have been invoked to describe this disparate group: simple bodied organisms, reproductive propagules lacking well-developed structures, as in flowers or cones, mostly aquatic, and generally photosynthetic. Yet, because the algae are polyphyletic and arising from various ancient ancestors, there are myriad exceptions to these characters as well as many taxa that fit these descriptions.
In reality, the tie that binds all algae together is a concept known as the 'serial endosymbiosis hypothesis' (Bermudes & Margulis 1985, Bhattacharya 2000), which suggests that the commonality between all algae is that, at one or more points in their lineage, they acquired photosynthetic abilities through either the uptake of a photoautotrophic bacteria (known as a primary endosymbiotic event), or through the uptake of a heterotroph that had previously taken up a photoautotrophic bacteria (a secondary endosymbiotic event). A current hot topic in algal phylogeny is determining whether there was one or two primary endosymbiotic events that give rise to red and green algae (e.g. Moreira et al. 2000). It is generally accepted that there were multiple secondary endosymbiotic events. With the advent of molecular tools, algal systematics is in the midst of a substantial re-working of these and other questions.

Small Perennial Kelp (Macrocystis pyrifera), photo by Michael Hawkes.
Molecular tools have also provided the opportunity to examine relationships between algae and land plants (Bhattacharya & Medlin 1998). Mounting evidence suggests that land plants are derived from an ancestor of the green algal order Charales (Michler & Churchill 1985, Surek et al. 1994). Land plants are excluded from algae because they do not conform to the character set described above; these plants have complex bodies, typically with flowers or cones, and are normally terrestrial.
The study of algae is called phycology, from the Greek root phykos meaning algae. Algae are a vast group including many classes of unicellular and multicellular organisms. Below, I focus on the multicellular macrophytes, and in particular, the benthic marine macrophytes that most refer to as the seaweeds.
What are Seaweeds?
Seaweeds are a macroscopic subset of marine algae (as opposed to the microscopic subset known as phytoplankton). The west coast of British Columbia has a diverse flora and is home arguably to the widest variety of kelp (Order Laminariales) in the world.
Which photosynthetic marine organisms are not seaweeds?
There are numerous marine macrophytes and other marine organisms with photosynthetic
endosymbionts that are not considered seaweeds. To reduce confusion, I briefly describe
them here:
Seagrasses (Zostera spp.) and surfgrasses (Phyllospadix spp.): These genera are in fact angiosperms that have moved into the marine environment. Although they are photosynthetic marine macrophytes, they reproduce using flowers and seeds. Both genera are important marine plants and are hosts to various epiphytic marine algae, but are not themselves seaweeds.
Marine lichens (e.g. Verrucaria spp.) Although lichens are a symbiosis between
cyanobacteria (an alga) and a fungus, and marine lichens do exist, these are not
considered seaweeds.
Corals and sea anemones: corals are actually colonial animals, although they derive some energy from algal symbionts. Sea anemones also have endosymbiotic algae (which in some cases give rise to the coloration of the anemone), but sea anemones are also animals.

Lessoniopsis littoralis, Constantinea simplex, Corallina vancouveriensis,
Phyllospadix scouleri, photo by Colin Bates
Major groups of seaweeds
The remaining photosynthetic macrophytes are the seaweeds. The seaweeds are split into
three major groups, originally delineated by their pigment complement (and hence color):
the green algae (Division Chlorophyta), the brown algae (Class Phaeophyceae, Division
Ochrophyta), and the red algae (Division Rhodophyta). I will refer to them henceforth as
the reds, greens, and browns.
Green seaweeds
The greens are often a brilliant grassy green, although some like Codium setchellii are so
dark green that they seem black in color. The bright green color stems from a lack of
accessory pigments to mask the Chlorophyll a pigments used in photosynthesis. There are
many freshwater green algae, but some classes of greens are exclusively marine (e.g.
Ulvophyceae, which contains the genera Ulva & Enteromorpha). Green seaweeds come
in a wide variety of morphologies, ranging from unicells and filaments to blades and
fleshy thalloid forms.
In general, the greens are encountered in the intertidal zone and lower subtidal. The diversity of greens in the Northeast Pacific includes 117 taxa in 51 genera (Gabrielson et al. 2000). Common genera of green algae in British Columbia include: Ulva, Enteromorpha, Acrosiphonia, Cladophora, Codium, and Prasiola. I provide here a few notes about each:
Ulva is represented by four species in British Columbia. Ulva is a thin distromatic green
blade that is fast growing and weedy. You may confuse Ulva with numerous other less common
green blades such as Ulvaria, Monostroma, or Kornmannia. These latter three
genera are monostromatic green blades. The most common species of Ulva that you will
encounter is Ulva fenestrata (a.k.a. Ulva lactuca) , which, as the name implies, has little holes or ‘windows’.
Enteromorpha, containing seven species in the Northeast Pacific, has traditionally been
separated from Ulva based on differences in morphology, with the Enteromorpha exhibiting “tube-like” growth. However, recent molecular evidence (Hayden et al. 2003)
has demonstrated that Ulva and Enteromorpha are not genetically different enough to
support a separation of the two genera. As a result, species previously known as
Enteromorpha are now known as Ulva, (e.g. Enteromorpha intestinalis = Ulva
intestinalis).
Acrosiphonia is a branched filamentous green that, as a result of the presence of tiny
hooked filamentous branches, grows in clumps that look like green dreadlocks.
Consequently, the common name for the locally abundant species Acrosiphonia coalita (a.k.a. Acrosiphonia mertensii) is
“Witches Hair”. Interestingly, part of the Acrosiphonia’s life history is spent as an
endophyte inside certain red algal species (Sussmann & DeWreede 2001) like Mazzaella
splendens and the ‘Petrocelis’ phase of Mastocarpus (see the section about red seaweeds
below).
Cladophora is a genus represented by 8 species in the Northeast Pacific. Cladophora is superficially similar to Acrosiphonia, but has much shorter filaments and lacks the hooked branches. As such, patches of Cladophora look instead like tufts of moss in the intertidal.
Codium is locally represented by three species, but two are common: Codium setchellii and Codium fragile. This is an interesting genus because the cells of Codium are
‘coenocytic’, meaning that they lack cross-walls in their cells. As a result, a Codium
fragile individual measuring up to 25cm in length can be composed of a single
multinucleate cell! Further, a subspecies called Codium fragile ssp. tomentosoides is a
highly successful invasive species recently introduced on the east coast of Canada
(Garbary et al. 1997).
Brown seaweeds
The diversity of brown algae in the Northeast Pacific includes 143 taxa in 66 genera
(Gabrielson et al. 2000). The browns are typically brownish in color due to the presence
of fucoxanthin & beta-carotene pigments, as well as chlorophyll a & c. The browns are
exclusively marine and range from microscopic filaments to the largest algae known.
Certain kelps can grow up to 50m in length, and other kelps can live up to 15 years.
Brown seaweeds make up the majority of the biomass in the intertidal and upper subtidal zones in British Columbia. Species from two orders of brown algae are particularly abundant: the kelps (Order Laminariales) and the rockweeds: (Order Fucales). These two groups are economically and ecologically very important.
Economically, kelps and rockweeds are harvested in their raw form for food products, cosmetic & luxury spa items, and fertilizers. As well, through processing, their cell wall components (alginic acids and polysaccharides) can be removed and used as emulsifiers, anticoagulants, and in the production of textiles and rubbers.
Ecologically, kelps and rockweeds are major habitat providers and nursery environments for fish, invertebrates, and for some other algae. Their extensive biomass provides a large amount of primary productivity oxygen to nearshore food webs. Further, nearshore kelp beds are useful for protecting shorelines from erosion by decreasing the impacts of water motion and storms. Clearly the browns are an important group of seaweeds.
The fucoids are represented in British Columbia by four genera: Fucus, Sargassum, Cystoseira, and Pelvetiopsis. Fucus is likely the most conspicuous seaweed in British Columbia, represented by two species: Fucus gardneri and Fucus spiralis. Sargassum is represented by one species, S. muticum, which is an introduced species, reportedly entering British Columbia prior to 1940 as a byproduct of importing oyster spat from its native country of Japan (Scagel 1956). To keep the oyster spat moist and cool, S. muticum was used as wrap for transport. Upon arrival, the Sargassum was presumably dumped into the British Columbia waters. Cystoseira is a less common alga that resembles Sargassum, and Pelvetiopsis limitata, which looks much like juvenile Fucus that lacks a midrib, is quite common in the upper intertidal of semi wave exposed British Columbia shores.
The diversity of kelps in British Columbia is unparalleled. Over 30 species of these beautiful brown algae can be found in most outer coast rocky habitats, and they range from the mid intertidal into the subtidal. The most conspicuous are the kelp forests composed of the giant kelp, Macrocystis integrifolia, and the bull-whip kelp, Nereocystis luetkeana. Among the most unique of the kelp is the sea palm, Postelsia almaeformis, found only on the most wave-battered shores. Its rarity and palm-tree like appearance makes it a rare treat for phycologists and others to view. In general, the kelp are fascinating and I often refer to them as ‘charismatic megaflora’.
Aside from the rockweeds and kelp, there are many other interesting brown algae to find on British Columbia shores. Of note are the bizarrely convoluted and brain-like Leathesia difformis, the ‘sausage-weed’ Scytosipon simplicissimus, and the bracket fungus like Ralfsia fungiformis. Also, a particularly interesting genus is Desmarestia, the acid seaweeds. Some species have an anti-herbivory adaptation that involves the release of sulfuric acid (pH of approximately 2; the same as your stomach!) when it is disturbed or exposed for air for too long. You may see it digesting itself after being swept onto a shore after a storm. If you are collecting seaweeds, make sure to put this one into its own bag to prevent it from bleaching out and digesting all of your other collections.
Red seaweeds
The red algae are the most diverse group of seaweeds in the Northeast Pacific, with 373
taxa across 162 genera (Gabrielson et al. 2000). The majority of red algae in British
Columbia are marine, although some genera (e.g. Batrachospermum) are found in
freshwater environments. The name ‘red algae’ can be misleading in this group because
although reds can indeed be red, they can also be green, violet, purple, yellow, brown,
pink, black, and even iridescent! This range of colors arises from varying amounts of
accessory pigments like phycoerythrin (which leads to a red color), phycocyanin (bluish
coloration), as well as violoxanthin and beta-carotene. Red algae have very complex and
interesting life cycles, and they lack flagellated stages in any part of their life history.
Reds are found from the intertidal to the deep limits of the photic zone, and display a
wide variety of morphologies including unicells, filaments, crusts, sacs, blades, and finely
branched forms.
The red algae are economically a very important group. Directly or indirectly (through
extraction of byproducts), red algae are a part of daily life for many North Americans,
whether they know it or not! Red algae are important as food, for compounds made with
their cell wall components, and for their potential as sources of natural chemicals.
Food: Many different red algae are utilized for food. Most familiar is ‘nori’ (Porphyra
yezoensis) which is used to make sushi rolls. Nori is farmed extensively in Asia, and an
unsuccessful attempt was made to farm nori in Washington State in the 1980’s. On the
east coast of Canada, two reds are very important as food. Irish Moss (Chondrus crispus)
is harvested in Prince Edward Island, and Bay of Fundy Dulse (Palmaria palmata) is
harvested on Grand Manan Island, New Brunswick.
Cell wall components: Red algal cell wall components, in particular agars and
carageenans, are used in cosmetics, food preparations, and biomedical and biotechnology
research. Ice cream contains red algal byproducts to emulsify the milk and water
products. Toothpaste has red algal byproducts included to keep the calcium carbonate
component mixed with the aqueous component. So, it seems that red algal byproducts
play an integral role in both the obtaining and preventing of tooth decay!
Natural Chemicals: Red algal species are being intensively surveyed for medicinal
properties and antiviral agents. Evidence exists to suggest that chemicals from red algae
can help to fight viral infections (Richards et al. 1978) like herpes (Dieg et al. 1974) and
HIV (Luescher-Mattli 2003).
Common reds along the BC coast include: Mazzaella splendens, Porphyra spp.,
Chondracanthus exasperatus, Microcladia coulteri, Mastocarpus spp., Prionitis
lanceolata, and various Coralline algae.
Mazzaella splendens is a common red bladed alga in British Columbia, and it can be
recognized by its iridescent sheen in the lower intertidal and in tidepools. This sheen is
caused by the same phenomenon that gives rise to iridescence when you pour oil on a
puddle: multiple layers (of cuticle in the case of Mazzaella) cause light to be
differentially refracted, giving the multiple colors of reflected light.
Chondracanthus exasperatus is commonly known as the “Turkish Towel”, and is a red
blade distinguished by its very bumpy (or ‘papillated’) texture. It also grows in the lower
intertidal, although you can usually find blades washed up on the beach after a storm. As
its name implies, this species is excellent for use in the shower, a natural exfoliating
cloth! You can usually use a blade for two or three days before it starts to disintegrate.
Microcladia coulteri is a finely branched and beautiful species. It is generally found as an
epiphyte (a plant growing on another plant) on M. splendens and C. exasperatus. M.
coulteri is perfect for pressing onto cardstock to make cards or art. Only take as much as
you will use though!
Mastocarpus papillatus is a common alga with two growth forms that you might not
think belong to the same species. The upright form (known commonly as the “Turkish
Washcloth”, has traditionally been associated with the genus Mastocarpus, and it is a
small and typically looks like a bifurcating dark brown blade with papillations. However,
the alternate phase is a reddish brown crustose phase that looks like tar on the rocks! For
a long time this phase was thought to belong to totally separate species known as
Petrocelis mittendorfi. Culture experiments demonstrated that these two phases were
actually part of the life cycle of one species. Hence, we refer to the crust as the
‘Petrocelis phase’ of the Mastocarpus life history.
Prionitis lanceolata (a.k.a.
Grateloupia americana) is known as ‘bleach weed’, and it grows in the lower intertidal zone.
This name stems from the scent of bromine that young P. lanceolata plants emit.
Presumably, this bromine acts as an antiherbivore chemical defense.
The coralline algae (Order Corallinales) have a unique adaptation to herbivory. This
group incorporates calcium carbonate into its cell walls, resulting in a very hard skeleton.
Both crustose forms and upright geniculate forms can be found in the Northeast Pacific,
and you can identify them by their brilliant pink color.
The reds are among the most beautiful of the algae, but usually they are the most underappreciated
because they have complex life cycles and they can be difficult to identify at
first. However, I assure you that if you spend some time getting to know the reds that
they will be your favorite group of seaweeds!
Concluding remarks
I hope that after reading this, you are left with the impression that marine algae are
important, interesting, and integral components of the coastal ecosystem. True, there are
some nuisance algae, but the majority are an unimposing lot, simply ‘going with the
flow’ of the waxing and waning tides. Next time you are on the rocky shore, take some
time to discover these natural wonders!
How do I learn more about seaweeds?
Phycological resources are abundant if you look in the right place. Here I list a number of
books, websites, journals, and forums that are particularly relevant to the seaweeds of the
Northeast Pacific. For those who are serious about getting a hands-on treatment, there are
a number of phycology immersion courses that one can take at the Bamfield Marine
Sciences Centre (www.bms.bc.ca) on the west coast of Vancouver Island.
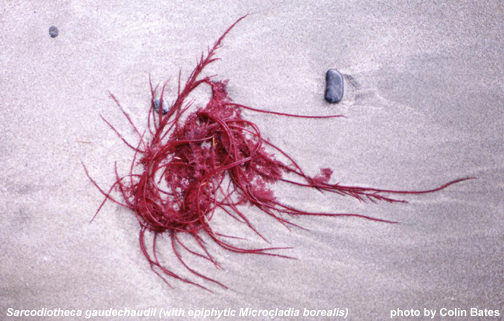
Sarcodiotheca gaudichaudii, photo by Colin Bates
Key References
Druehl L.D. (2001) Pacific Seaweeds. Harbour Publishing. 192p.
Gabrielson, Paul W., Thomas B. Widdowson, Sandra C. Lindstrom, Michael W. Hawkes and Robert F. Scagel. 2000. Keys to the Benthic Marine Algae and Seagrasses of British Columbia, Southeast Alaska, Washington and Oregon. Phycological Contribution Number 5. Department of Botany, University of British Columbia. 2000
Hawkes, MW. 1994. Benthic Marine Algal Flora (Seaweeds) of British Columbia: Diversity and Conservation Status. Chap. 11 In: Biodiversity in British Columbia: Our Changing Environment, edited by E. McCullum & L. Harding. Environment Canada: Vancouver.
Lindstrom, SC, JL Olsen, & WT Stam. 1997. Postglacial recolonization and the biogeography of Palmaria mollis (Rhodophyta) along the northeast Pacific coast. Can. J. Bot. 75: 1887-1896.
Lindstrom, SC, JP Houghton & DC Lees. 1999. Intertidal macroalgal community structure in southwestern Prince William Sound, Alaska. Bot. Mar. 42: 265-280.
Nelson, WA., GA Knight, & MW Hawkes. 1998. Porphyra lilliputiana sp. nov. (Bangiales, Rhodophyta) a diminutive New Zealand endemic with novel reproductive biology. Phycological Research 46: 57-61.
O'Clair, R. M., and S. C. Lindstrom. 2000. North Pacific Seaweeds. Plant Press, Juneau, xii + 162 pp. + 16 color plates.
Scagel, RF, PW Gabrielson, DJ Garbary, L Golden, MW Hawkes, SC Lindstrom, JC Oliveira and TB Widdowson. 1993 (reprint and revision of 1989 edition). A Synopsis of the Benthic Marine Algae of British Columbia, Southeast Alaska, Washington and Oregon. Phycological Contribution no. 3: vi + 535 pp. Dept. of Botany, University of British Columbia: Vancouver.
Scagel, R. F., P. W. Gabrielson, D. J. Garbary, L. Golden, M. W. Hawkes, S. C. Lindstrom, J. C. Oliveira and T. B. Widdowson. 1989 [ Reprinted 1993 with minor changes]. A Synopsis of the Benthic Marine Algae of British Columbia, Southeast Alaska, Washington and Oregon. Phycological Contribution Number 3. Department of Botany, University of British Columbia.

Sea Sac (Halosaccion glandiforme), photo by Colin Bates.
Journals
Bermudes D, and L Margulis (1985) Symbiosis as a Mechanism of Evolution: Status of
the Symbiosis Theory. Symbiosis 1: 101-124.
Bhattacharya D (2000) A molecular phylogenetic perspective on the primary and
secondary endosymbiotic origins of algal plastids. Algae 15:1-5.
Bhattacharya D, and L Medlin (1998) Algal phylogeny and the origin of land plants.
Plant Physiol. 116: 9-15.
D Moreira H, Le Guyadev, and H Phillipe (2000) The origin of red algae and the
evolution of chloroplasts. Nature 405: 69-72.
Dieg et al. (1974) "Inhibition of herpes virus replication by marine algae extracts,"
Anitimicrb. Ag. Chemother. 6:524-525.
Gabrielson PW, Widdowson TB, Lindstrom SC, Hawkes MW, and Scagel, RF (2000)
Keys to the Benthic Marine Algae and Seagrasses of British Columbia, Southeast Alaska,
Washington and Oregon. Phycological Contribution #5, University of British Columbia,
Department of Botany. 189p.
Garbary DJ, Vandermeulen H, and Kim KY (1997) Codium fragile ssp. tomentosoides (Chlorophyta) invades the Gulf of St. Lawrence, Atlantic Canada. Botanica Marina.
40:(6) 537-540.
Graham L and Wilcox (2000) Algae. Prentice Hall.
Hayden HS, Blomster J, Maggs C, Silva PC, Stanhope MJ, and Waaland JR (2003)
Linnaeus was right all along: Ulva and Enteromorpha are not distinct genera. Eur. J.
Phycol. 38: 277-294.
Luescher-Mattli M (2003) Algae, A Possible Source for New Drugs in the Treatment of
HIV and Other Viral Diseases. Current Medicinal Chemistry - Anti-Infective Agents 2:
219-225.
Meinesz A (1999) Killer Algae. University of Chicago Press. 376 p.
Mishler BD & Churchill SP (1985) Transition to a land flora: phylogenetic relationships
of the green algae and bryophytes. Cladistics 1:305-28.
Richards et al. (1978), "Antiviral activity of extracts from marine algae," Antimicrob.
Agents Chemother. 14: 24-30.
Scagel RF (1956). Introduction of a Japanese alga, Sargassum muticum, into the
Northeast Pacific. Fisheries Research Papers 1(4): 49-58.
Surek B, U Beemelmenns, M Melkonian, and D Bhattacharya (1994). Ribosomal RNA
sequence comparisons demonstrate an evolutionary relationship between Zygnematales
and charophytes. Pl. Syst. Evol. 191:171-181.
Sussmann AV and RE DeWreede (2001) Life history of Acrosiphonia (Codiolales,
Chlorophyta) in southwestern British Columbia, Canada. American Journal of Botany 88: 1535-1544.
Please cite this page as: Bates, Colin. 2004. An Introduction to the Algae of British Columbia. In: Klinkenberg, Brian.(Editor) 2004. E-Flora BC: Atlas of the Plants of British Columbia [www.eflora.bc.ca]. Lab for Advanced Spatial Analysis, Department of Geography, University of British Columbia, Vancouver. |
Recommended citation: Author, date, page title. In: Klinkenberg, Brian. (Editor) 2021. E-Flora BC: Electronic Atlas of the Flora of British Columbia [eflora.bc.ca]. Lab for Advanced Spatial Analysis, Department of Geography, University of British Columbia, Vancouver. [Date Accessed]
E-Flora BC: An initiative of the Spatial Data Lab, Department of Geography UBC, and the UBC Herbarium.
© Copyright 2021 E-Flora BC.